- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
IV Workflow for PN Compounding Optimization
The use of parenteral nutrition (PN) is not uncommon in the United States. The 2022 National Inpatient Survey data shows that PN preparations were prescribed in over 290,000 hospital stays for a variety of indications, with roughly 43% of those preparations indicated for use in pediatric and newborn patients.1 Despite their common use, there is the potential for an increased risk of harm to patients affected by an error related to a faulty PN bag. Accordingly, the Institute for Safe Medication Practices (ISMP) classifies PN preparations as high-alert medications and urges facilities using these products to implement strategies to mitigate the risk of error associated with their use.2
Additionally, the American Society for Parenteral and Enteral Nut-rition (ASPEN) has established safe practices for parenteral nutrition guidelines, recommending best practices related to the prescribing, preparation, and administration of PN across multiple health care settings.3 In the 2013 revision of these guidelines, ASPEN recommends that organizations “develop a strategic plan for implementation of automation and technology for sterile products service” as it relates to PN production.3 Utilizing an IV workflow system (IVWFS) for PN compounding offers well-documented safety benefits that can address the safety concerns associated with PN use, as well as delivering additional benefits that are not as frequently discussed.
AdventHealth Orlando, which corporately represents the Central Florida Division-South of AdventHealth, is a large, community-based health care system located in Central Florida. The division encompasses nine hospitals ranging in size from 120 to 1300 beds and includes a 250-bed pediatric hospital as well as seven freestanding emergency departments. The division also operates a centralized distribution pharmacy, Central Fill Pharmacy (CFP), that provides daily automated dispensing cabinet (ADC) replenishment to all facilities. Since 2019, CFP has provided insourced PN compounding for all of the hospital sites in the Central Flo-rida Division-South. CFP produces an average of 60 PN preparations each day, including standardized formulations and patient-specific customized formulations. Roughly half of these PN preparations are for pediatric or neonatal patients.
IV Workflow System Implementation
In 2020, AdventHealth began rolling implementations of an IVWFS across its hospital and infusion center locations. Adding the IVWFS at CFP seemed a natural next step to achieve greater quality assurance, enhance safety features, and streamline workflow processes.
PN Workflow
At each hospital campus, physicians consult clinical pharmacists to assess patient PN requirements, as it falls under the clinical pharmacist’s scope of practice to order standard or custom formulated PN. The clinical pharmacist enters the order into the electronic health record (EHR) and creates a PN order entry application, both of which are double-checked by a second pharmacist at their facility before being marked as complete and sent to CFP for compounding.
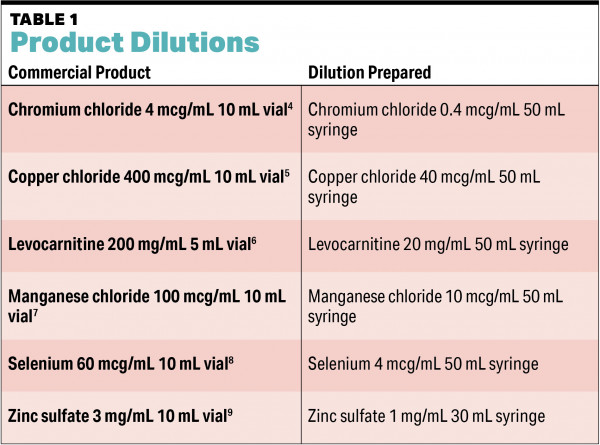
All of the PN compounding at CFP is directly overseen by a pharmacist and performed by a single pharmacy technician. First, the pharmacist uses quantities from the previous day to gauge the necessary components, quantities, and supplies for the day. Next, each product is wiped down before being transferred into the cleanroom. Once garbed and inside the room, the technician starts their shift by making stock syringes. These include products that are diluted before use as well as products pooled into larger volume stock syringes (see TABLE 1 and TABLE 2). Once the stock syringes are prepared, the technician sets up the automated compounding device (ACD). Finally, the pharmacist enters the cleanroom to visually inspect the ACD and confirm it is set up appropriately before compounding begins.

IVWFS Realized Benefits
Product Labeling Enhancement
Prior to IVWFS implementation, all PN bags had two product labels. The first label was generated by the EHR to ensure PN doses could be documented through bar code medication administration (BCMA), and the other was generated by the PN order entry system to communicate the required bag components and volumes to the ACD. This process was riddled with opportunities for human error. One issue was nurses unfamiliar with PN confusing which label was supposed to be scanned for use with the EHR. Another drawback with this process was the extra attention required to ensure that the correct EHR label was applied appropriately to the corresponding compounded bag, matching the ACD label. Instances of mislabeling resulted in the dose being held, since the nurse and frontline pharmacy staff were unable to confidently resolve the discrepancy between the ACD compounded label and EHR ordered label.
As part of the IVWFS implementation, the pharmacy informatics team at AdventHealth-Orlando was able to work with the vendor to consolidate the two product labels into a single label (see FIGURE 1). The team built an excellent rapport with the vendor through the previous implementations and was able to leverage those relationships to work together on a single label solution. The new label contains a linear bar code that the ACD can read to drive compounding as well as a two-dimensional bar code that only appears on the final verified label for BCMA. This collaboration and result greatly reduced the risk of inappro-priate scanning and incorrect labeling errors.
Streamlined Documentation
Before the IVWFS was in place, all record keeping information was manually entered into a separate software system. This encompassed all products and supplies used to compound PN bags, including product descriptions, NDC numbers, lot numbers, expiration dates, beyond use dates (BUDs), and volumes used. Falling under the pharmacist’s purview, manual documentation was time-consuming and inefficient. Furthermore, because the process for entering the information varied from one pharmacist to another, the documentation was not always completed in a timely manner.
With IVWFS, all documentation is performed in real time at the point of preparation, and much of the information is automatically documented by scanning a bar code. When the technician prepares stock solutions, each product used is scanned to capture the NDC and is assigned a product specific BUD. Next, the system prompts the user to enter lot numbers, expiration dates, and quantities of each ingredient being used. Since the communication from the PN order entry system flows through the IVWFS, the volumes used in each order are also captured and archived. Documenting these steps cannot be skipped using the IVWFS, creating assurance that these values are documented at the time of preparation. Additionally, shifting this responsibility to the compounding technician frees up the pharmacist.
Workflow Management
Another benefit of using an IVWFS in PN compounding is improved workflow management. Previously, certain aspects of the PN compounding process could vary depending on the pharmacist or technician working. One example was the prepping process for the following shift: some pharmacists would leave handwritten notes for the incoming staff to give insight as to the anticipated number of dilutions or stock syringes that to prepare, while others would not. With the IVWFS, the pharmacist can proactively queue up the necessary dilutions and stocks for the next day, allowing the technician to hit the ground running with tasks waiting for them as soon as they garb and enter the cleanroom.
The IVWFS also provides the pharmacist with a streamlined process to triage the order in which bags are prepared. At AdventHealth, PN bags are picked up and delivered to the hospital campus every afternoon via an internal courier service whose route is based on geographic location. Before the IVWFS implementation, the technician running the ACD had little direction regarding bag prioritization, as labels were printed and given to the technician in the order that they were entered. Having to rely on first printed/first compounded without regard to delivery schedules had the potential to cause distribution delays. Now, using the IVWFS, the pharmacist can easily and accurately prioritize the order in which bags are compounded to ensure orders are completed in sync with the established delivery schedule.
Remote Verification
A commonly recognized benefit of using IVWFS is the ability to verify orders remotely. The technician takes multiple, high quality photos of the ingredients, the actual volumes used, and the final product. This information, combined with the data entered before compounding, allows the pharmacist to verify orders outside of the cleanroom. Previously, the pharmacist at CFP had to be inside the cleanroom for the duration of compounding, as they were required to verify all manual additions to PN bags that occurred outside of the ACD. This made it difficult for the pharmacist to communicate with anyone in the facility concerning inventory issues that might arise inside the cleanroom. Furthermore, it made communication with campus-based clinical pharmacists who might have a question about a particular PN preparation, a change to an existing order, or an order discontinuation even more difficult. In addition, the previous workflow required a third person stationed outside the cleanroom to prepare completed bags for distribution.
Now that remote verification is available, the pharmacist can be stationed outside of the cleanroom for most of the day. They can readily communicate with the clinical pharmacists at the various campuses, troubleshoot any issues that might be occurring, supply additional items that are needed inside the clean room, and organize bags for distribution. At times, they are available to help with other operational tasks in the centralized pharmacy.
Reporting Capabilities
For the insourced operation at AdventHealth, the central fill facility purchases all the drugs and supplies needed to compound PN preparations. At the end of each month, the cost of these components is billed to the respective campuses via an internal departmental transfer (IDT). Previously, this documentation task was arduous and fell on the compounding pharmacist and leadership team to complete. Each day, the pharmacists manually tracked the ingredients used in an Excel spreadsheet. The ingredient costs were then added up and divided among the campuses based on their overall PN bag counts.
With the addition of the IVWFS, the pharmacy informatics team, again leveraging their relationship with the vendor, was able to develop custom reports to capture ingredient use by campus (see FIGURE 2). While obstacles remain in determining the best way to bill waste from discontinued bags or partial vial usage, the new reporting features provide a mechanism for more accurate charge transfers.
Implementation Tips
Many contributing factors play a role in the success of a significant process improvement initiative like IVWFS implementation. To achieve the benefits described herein, there are three key proponents. Identify and communicate your goals to all key stakeholders. Keeping the “why” at the forefront of the change (eg, incorporating automation and technology to achieve safety enhancements and a more efficient workflow) helps garner support for the changes being made.
Equally important is establishing a collaborative relationship with the vendor. The rapport our pharmacy informatics team was able to create with the IVWFS vendor greatly contributed to the success of this implementation. Through collaboration, they were able to find creative solutions to better meet our operational needs.
Finally, take care to ensure appropriate go-live support. With IVWFS go-lives, team members are commonly concerned that the new process will take much longer than the previous operations. Having both the vendor and a thoroughly trained pharmacy informatics team onsite to work shoulder-to-shoulder with the frontline compounding staff put our team at ease knowing they had resources available for any questions or concerns with the new system.
Conclusion
Multiple organizations, including ASPEN and ISMP, have taken clear stances on the importance of promoting the safest methods possible for ordering, compounding, and administering PN admixtures. Utilizing an IVWFS in PN compounding can help to increase quality assurance, streamline workflow, and ensure the compounding tasks are being completed safely.
 Abbi J. Rowe, PharmD, MHA, 340B ACE, is the director of pharmacy at AdventHealth Central Fill Pharmacy in Orlando, Florida. She received her doctor of pharmacy degree from Mercer University, completed two years of pharmacy residency training with a focus in Health System Pharmacy Administration at AdventHealth Orlando, and obtained her Master of Healthcare Administration from Adventist University of Health Sciences.
Abbi J. Rowe, PharmD, MHA, 340B ACE, is the director of pharmacy at AdventHealth Central Fill Pharmacy in Orlando, Florida. She received her doctor of pharmacy degree from Mercer University, completed two years of pharmacy residency training with a focus in Health System Pharmacy Administration at AdventHealth Orlando, and obtained her Master of Healthcare Administration from Adventist University of Health Sciences.
 Hiren M. Shah, PharmD, BCPS, is a pharmacy informatics specialist at AdventHealth Orlando. Hiren completed his PharmD at the University of Florida, PGY-1 pharmacy residency training at Lakeland Regional Health, and PGY-2 pharmacy informatics residency training at AdventHealth Orlando.
Hiren M. Shah, PharmD, BCPS, is a pharmacy informatics specialist at AdventHealth Orlando. Hiren completed his PharmD at the University of Florida, PGY-1 pharmacy residency training at Lakeland Regional Health, and PGY-2 pharmacy informatics residency training at AdventHealth Orlando.
 Dom Bracero, MBA, RPh, CPh, BCSCP, is the executive director of supply chain and logistics at AdventHealth Central Florida Division South. He received his bachelor of science in pharmacy from St. John’s University in Queens, New York. He earned his MBA degree from Ashford University in Clinton, Iowa.
Dom Bracero, MBA, RPh, CPh, BCSCP, is the executive director of supply chain and logistics at AdventHealth Central Florida Division South. He received his bachelor of science in pharmacy from St. John’s University in Queens, New York. He earned his MBA degree from Ashford University in Clinton, Iowa.
References
- Blackmer A. What Is Parenteral Nutrition. American Society for Parenteral and Enteral Nutrition (ASPEN). 2022. Accessed October 4, 2022. https://www.nutritioncare.org/About_Clinical_ Nutrition/What_is_Parenteral_Nutrition/
- Institute for Safe Medication Practices. ISMP List of High-Alert Medications. ISMP. October 2018. Accessed October 4, 2022. https://www.ismp.org/sites/default/files/attachments/2018-10/highAlert2018new-Oct2018-v1.pdf
- Ayers P, Adams S, Boullata J, et al. ASPEN Parenteral Nutrition Safety Consensus
Recommendations. J Parenter Enteral Nutr. 2014;38(3):296-333. - Chromium [package insert]. Lake Forest, IL: Hospira, Inc. https://www.pfizer hospitalus.com/products. Published April 2021. Accessed January 11, 2023.
- Copper [package insert]. Lake Forest, IL: Hospira, Inc. https://www.pfizer hospitalus.com/products. Published April 2021. Accessed January 11, 2023.
- Carnitor [package insert]. Gaithersburg, MD: Leadiant Biosciences, Inc. https:// leadiant.com/products/. Published April 2021. Accessed January 11, 2023.
- Manganese [package insert]. Lake Forest, IL: Hospira, Inc. https://www.pfizer hospitalus.com/products. Published April 2018. Accessed January 11, 2023.
- Selenious acid [package insert]. Shirley, NY: American Regent, Inc. https://american regent.com/our-products/. Published August 2021. Accessed January 11, 2023.
- Zinc sulfate [package insert]. Shirley, NY: American Regent, Inc. https://american regent.com/our-products/. Published October 2020. Accessed January 11, 2023.
Like what you've read? Please log in or create a free account to enjoy more of what www.pppmag.com has to offer.










