- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
Address PN Shortages with MCB-PN

Shortages abound in the health care industry of late, and parenteral nutrition (PN) is one of the many areas affected by this challenge. Components and supplies for PN are not always readily available, leading institutions to look for alternatives to customized, compounded products. The utilization of multi-chamber bag PN (MCB-PN) is key in addressing this need.
MCB-PN is a standardized, commercially available PN product that can provide a complete nutrition support regimen when appropriate components are added and it is prescribed at a correct rate. These products can be used in place of customized PN in appropriate patient populations to help address drug and supply shortages. However, it is important to avoid using the term premixed when referring to MCB-PN, as these products require mixing and the addition of additives.
Available Products
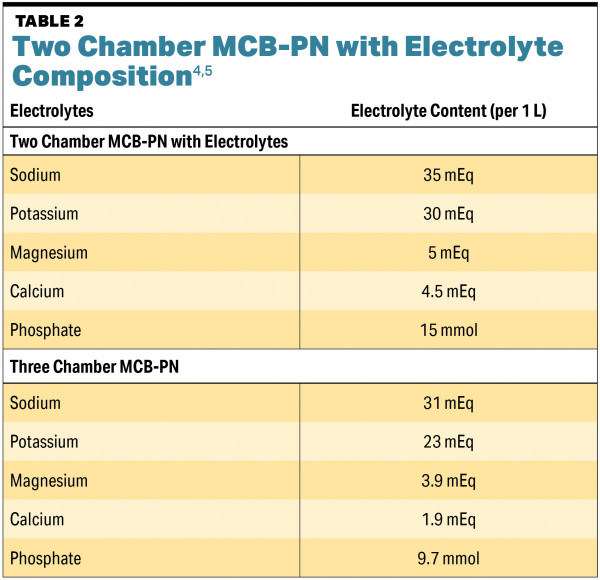
In the United States, there are currently two MCB-PN products. There is a two chamber (dextrose and amino acid) product that comes with or without electrolytes, and there is a three chamber product (lipid injectable emulsion (ILE), dextrose, and amino acid) that comes with electrolytes. See TABLE 1 for a comparison of the composition of the current MCB-PN products in the United States.1,2,6-8
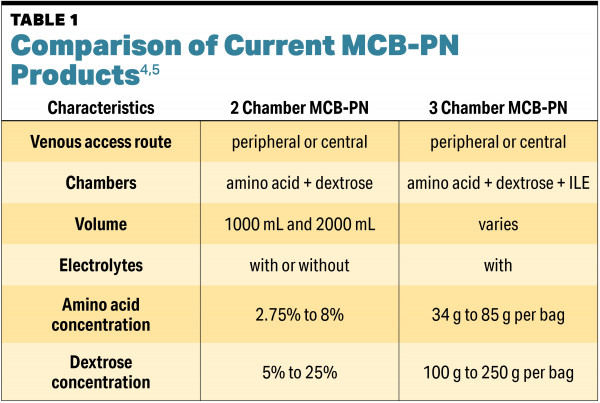
Currently, the three chamber MCB-PN in the US comes with electrolytes only. Despite current recommendations from ASPEN, there is reporting of electrolytes in per liter amounts and macronutrients as final percentages in the two chamber MCB-PN. To compare the electrolyte content of the two products, TABLE 2 shows the amount of electrolytes in per-liter amounts. However, be advised that current ASPEN recommendations state that these amounts should be reported in electrolyte and macronutrient per day.1,2,6-8
Advantages and Disadvantages
The utilization of MCB-PN carries several potential advantages. MCB-PN requires less manipulation than traditional compounded PN; nevertheless, MCB-PN activation and the addition of additives should still be conducted in a sterile environment. With MCB-PN, there is the possibility of mitigating medication and supply shortages if it can meet patient needs. There are also reduced time and staffing commitments when utilizing MCB-PN, creating the potential for cost and time savings.1,2,6-8
It is important to note that because these products contain a fixed amount of electrolytes, their utility for patients with large electrolyte requirements may be limited. Many patients will not receive a full bag of MCB-PN, so there is a potential for waste and erroneous administration when used for longer than 24 hours. Furthermore, when using a combination of MCB-PN and compounded PN, there is a possibility for errors resulting from the two separate methods of ordering and the different ways macronutrients and electrolytes are reported.1,2,6-8
Adult Utilization
The use of MCB-PN in the adult population has increased in response to shortages of PN components and supplies. MCB-PN may often be used in place of compounded/customized PN in appropriate patient populations and settings. Studies have shown that MCB-PN may offer some advantages over compounded PN such as reduced cost, decreased compounding time, reduced chance of error, and diminished incidence of bloodstream infections.1 It is important to note that MCB-PN may not be appropriate for all patients and may have additional costs associated with use.1
Successful utilization of MCB-PN requires careful assessment and evaluation by the clinician. Two recent articles provide excellent guidance for incorporating MCB-PN into practice.1,2 Hall addresses the safety, cost, and clinical considerations and utilizes a flow chart to assist in determining if MCB-PN is appropriate for the patient, while Nystrom and colleagues discuss strategies utilized by a nutrition support service during an amino acid shortage that limited the ability to compound PN.1,2 Both articles emphasize the need for patients to be metabolically stable in order to consider utilizing MCB-PN.
Adult Limitations
MCB-PN can provide the necessary macronutrients and electrolytes in select patients. Hall noted an increase in utilization of MCB-PN from 10% to 20% using a flow chart with criteria outlining appropriate selection. It should be noted that the majority of patients were non-critically ill.1 Nystrom et al reduced the use of compounded adult PN to 25% of pre-shortage practice by utilizing MCB-PN in appropriate patients.2
Organizations should review current PN formulations and indications to determine if the use of MCB-PN is appropriate. Circumstances in which the utilization of MCB-PN may not be optimal include:1,2
- PN duration >2 weeks
- Electrolyte abnormalities
- High output ostomy or fistula
- Obesity
- Renal failure (acute or chronic)
- Volume sensitivity
- Cardiopulmonary instability
- High protein requirements
Adult Safety Concerns
MCB-PN is considered a safe alternative to compounded PN for appropriate patients. These products should not be referred to as premixed because they require activation and intravenous multivitamins, and trace elements should be added before dispensing for administration. Activation and additions of essential components should be done in the pharmacy using aseptic technique.
Potential safety concerns associated with the use of MCB-PN include:
- Not being seen as PN, which could lead to issues such as refeeding syndrome overfeeding, underfeeding, and electrolyte derangements
- Not activated properly
- Allowed to hang over 24 hours, leading to an increased risk of infection
- Additions of additives that may alter the stability of the MCB-PN
- Addition of essential components omitted (MVI, trace elements)
- ILE not administered with use of 2 chamber MCB-PN leading to essential fatty acid deficiency
- Transition of care errors
- Use in the home environment with inadequate patient/caregiver education
Pediatric Utilization
Shortages of drugs and supplies needed for PN compounding have required nutrition support clinicians to think outside the box, including consideration of MCB-PN use in children. Currently, there are no MCB-PN products in the US specifically formulated for pediatrics, and there is limited data regarding MCB-PN use in children in the US. One 2015 US-based study concluded that with close monitoring, MCB-PN could provide a safe and effective option for pediatric patients requiring PN for short courses, especially during times of drug shortages.3
Despite these studies, available formulations are not well matched to the nutritional requirements of premature neonates and infants. Additionally, MCB-PN is unlikely to meet the needs of patients who are critically ill, those with electrolyte abnormalities, renal dysfunction, or high fluid or electrolyte needs. The best candidates are older pediatric patients, especially those with an anticipated short PN course who are in relatively stable condition. MCB-PN may be a short-term option for some children requiring long-term PN, including those on PN at home. However, shortages may necessitate MCB-PN consideration for patients who fall into less ideal categories.
Pediatric Limitations
Children have a wide range of nutritional needs based on age, weight, and clinical condition. Since there are no pediatric MCB-PN formulations, there are limitations and challenges with pediatric use. Daily total protein need is lower for children than adults, so the protein dose will usually dictate the maximum rate or volume of MCB-PN. However, because pediatric patients often require a higher glucose infusion rate, additional dextrose containing fluids in addition to the MCB-PN may be needed. Likewise, MCB-PN does not contain the amino acids cysteine or taurine, which are considered conditionally essential in neonates due to their immature metabolic systems.
Total fluids for each patient must be carefully considered as MCB-PN alone may not provide goal fluid amounts. Electrolyte provision, especially with regard to calcium and phosphorus required for bone building, may not match requirements in younger pediatric patients. It is possible to add electrolytes to MCB-PN with guidance from the manufacturer, but amounts that can be added are limited.
Three chamber MCB-PN contains lipid ILE and is not recommended for use in patients less than 2 years old, so a separate ILE infusion may be required.4 Further, it is important to remember that MCB-PN formulations do not contain multivitamin or trace elements, which should be added separately per recommended pediatric dosing.
Pediatric Safety Concerns
Pediatric dosing of MCB-PN is very complex due to the fixed ingredient ratios of available formulations. Pediatric specific rates included in prescribing information are based on protein dose and do not consider total carbohydrate, fluid, or electrolyte needs.5 Dosing of each of these components must be considered individually. The complexities of these regimens underline the importance of having clinicians trained in nutrition support involved in these patients’ care.
Soybean-oil based ILE, which is a component of three chamber MCB-PN, carries a black box warning of death in preterm infants due to pulmonary lipid accumulation. Special attention must be paid to ILE dosing and infusion rates if using three chamber MCB-PN in pediatric patients.
While additions to MCB-PN are possible, any addition must be evaluated for compatibility. An important example would be the addition of extra calcium and phosphorus to an MCB-PN. Specific recommendations are available from MCB-PN manufacturers and should be closely followed.
Clinicians monitoring pediatric patients on MCB-PN regimens should pay special attention to serum glucose—especially in neonates and infants—electrolytes, triglyceride (if ILE are included), and overall growth to ensure the adequacy of the chosen MCB-PN regimen.
Conclusion
Considering recent drug and supply shortages of TPN, it is important to look beyond traditional treatment options for patients requiring PN. MCB-PN can serve to help ensure patient access to care. When used correctly in eligible patients, this product can prevent treatment interruptions, with the added benefit of delivering potential cost and time savings.
References
- Hall JW. Safety, Cost and Clinical Considerations for the Use of Premixed Parenteral Nutrition. Nutr Clin Pract. 2015;30:325-330.
- Nystrom EM, Bergquist WJ, Wieruszewski PM, et al. Parenteral Nutrition Drug Shortages: A Single-Center Experience With Rapid Process Change. JPEN J Parenteral Enteral Nutr. 2019;43:583-590.
- Chhim RF, Crill CM. Premixed Parenteral Nutrition Solution Use in Children. J Pediatr Pharmacol Ther. 2015;20(5):378-384. doi:10.5863/1551-6776-20.5.378
- Kabiven Package Insert. Fresenius Kabi. Updated October 2106. Accessed June 8, 2022. https://e5e2j7i8.stackpathcdn.com/wp-content/uploads/2020/09/US-PH-Kabiven-FK-451206C-10-2016-PI.pdf
- Clinimix-E Package Insert. Baxter. Updated April 2021. Accessed June 8, 2022. www.baxterpi.com/pi-pdf/Clinimix_E_PI.pdf
- Ayers P, Adams S, Boullata J, et al. American Society for Parenteral and Enteral Nutrition. A.S.P.E.N. parenteral nutrition safety consensus recommendations. JPEN J Parenter Enteral Nutr. 2014 Mar-Apr;38(3):296-333.
- Boullata JI, Gilbert K, Sacks G, et al. American Society for Parenteral and Enteral Nutrition. A.S.P.E.N. clinical guidelines: parenteral nutrition ordering, order review, compounding, labeling, and dispensing. JPEN J Parenter Enteral Nutr. 2014 Mar-Apr;38(3):334-77
- Kochevar M, et al. A.S.P.E.N. Statement on parenteral nutrition standardization. JPEN 2007;31:441-8.
 Andrew Mays, PharmD, MBA, BCNSP, CNSC, FASPEN, is a clinical pharmacy specialist in nutrition support at the University of Mississippi Medical Center in Jackson, Mississippi. He is a clinical assistant professor at the University of Mississippi School of Pharmacy. Andrew received his PharmD and MBA from the University of Mississippi. He serves as the current chair for ASPEN’s Parenteral Nutrition Safety Committee.
Andrew Mays, PharmD, MBA, BCNSP, CNSC, FASPEN, is a clinical pharmacy specialist in nutrition support at the University of Mississippi Medical Center in Jackson, Mississippi. He is a clinical assistant professor at the University of Mississippi School of Pharmacy. Andrew received his PharmD and MBA from the University of Mississippi. He serves as the current chair for ASPEN’s Parenteral Nutrition Safety Committee.
 Phil Ayers, PharmD, BCNSP, FMSHP, FASHP, is chief, clinical pharmacy services, Baptist Medical Center, and associate clinical professor at the University of Mississippi School of Pharmacy. He received his BS in pharmacy and his PharmD from the University of Mississippi. Phil serves as secretary-treasurer for the board of directors of ASPEN, is the past chair of the ASPEN Parenteral Nutrition Committee, and is the chair of the USP Parenteral Nutrition Expert Panel.
Phil Ayers, PharmD, BCNSP, FMSHP, FASHP, is chief, clinical pharmacy services, Baptist Medical Center, and associate clinical professor at the University of Mississippi School of Pharmacy. He received his BS in pharmacy and his PharmD from the University of Mississippi. Phil serves as secretary-treasurer for the board of directors of ASPEN, is the past chair of the ASPEN Parenteral Nutrition Committee, and is the chair of the USP Parenteral Nutrition Expert Panel.
Margaret K. Murphy, PharmD, BCNSP, is a nutrition support pharmacist at Boston Children’s Hospital in Boston, Massachusetts. She received her PharmD from Wilkes University in Pennsylvania. Dr. Murphy is a member of the Home Parenteral Nutrition Team as well as the Center for Advanced Intestinal Rehabilitation. She also precepts pharmacy students as well as PGY-2 pharmacy residents.
Like what you've read? Please log in or create a free account to enjoy more of what www.pppmag.com has to offer.