- Show Menu
- Contact Us
- FAQs
- Reader Service
- Survey Data
- Survey Winners
- Testimonials
- Upcoming Events
- Webinars
- White Papers
Ensure Safe HD Handling
Q: How can hazardous drug handling practices be improved to ensure safety and compliance?
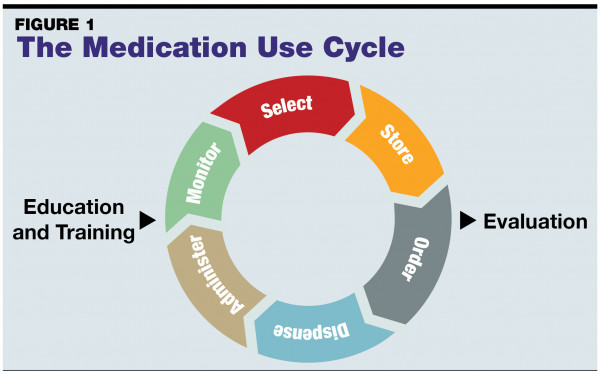
A: Organizations can improve hazardous drug (HD) handling safety by applying steps recognized as best practices throughout the medication use cycle (see FIGURE 1). Additionally, the National Institute of Occupational Safety and Health (NIOSH) defines the expected methods of risk mitigation in their Hierarchy of Controls, highlighting key strategies which include removal of the hazard, substituting an option posing less risk, the use of containment devices, administrative controls, and use of personal protective equipment (PPE). Removal of the hazard is an unlikely approach, since patients need to be treated with these agents that can be harmful to caregivers if not properly contained. Substitution is a rare approach, such as when a new dosage form or other improvement in the treatment is available.
Generally, the key approaches to workplace safety for health care personnel are use of containment devices, such as biological safety cabinets (BSCs), compounding aseptic containment isolators (CACIs), negative pressure cleanrooms, and closed system drug-transfer devices (CSTDs); use of PPE designed for protection from HDs; and the establishment and use of policies and procedures, which must include an assessment of risk.
Q: What is the value of conducting an assessment of risk?
A: Organizations need to review the HDs they handle in order to assess the risks posed to personnel. By creating an assessment of risk, pharmacy can triage risk and implement practices that differ from those dictated in USP <800> for those HDs that are not APIs or NIOSH Table 1 antineoplastics that require manipulation prior to administration. Pharmacy should begin with the list of hazardous drugs maintained by NIOSH, which must be used to create an assessment of risk as defined in USP <800>.1 The list indicates those drugs which pose risks to health care professionals when touched, inhaled, or injected. It is important to note that dermal absorption of the HDs is the most common way health care personnel are exposed.
Cytotoxic agents and any API HD on the NIOSH list that must be manipulated by personnel must follow all the containment practices (including the use of primary engineering controls, negative pressure rooms, and CSTDs) and work practices (such as training, material transfer, proper technique, and decontamination of surfaces) that are included in USP <800>. Some drugs (those that are neither API nor antineoplastics in Table 1 of the NIOSH list) and dosage forms (such as solid oral tablets) may be exempted by the organization if alternative strategies are identified and implemented. However, if an assessment of risk is not completed, all containment and work practices listed in USP <800> must be followed for all dosage forms of the HDs included on the NIOSH list.
Q: What are the key elements of an assessment of risk?
A: According to USP <800>, an assessment of risk must include the following: type of HD, dosage form, risk of exposure (as determined by the organization), type of packaging in which the HD is received, and what, if any, manipulation of the dosage form must be done prior to dispensing for patient use. The organization’s HD list and assessment of risk needs to include all HDs handled in the organization.
The following steps should be taken to create and maintain an assessment of risk:
- Download the most recent version of the NIOSH list of hazardous drugs, available online via the CDC.1
- Review purchase records for HDs that have been ordered for every area in the organization.
- Identify any HD API and HD antineoplastics in NIOSH Table 1 that must be compounded or otherwise manipulated prior to dispensing for patient use. These HDs need to be stored and compounded with all the containment and work practices identified in USP <800>. If refrigeration is needed, a separate refrigerator must be used and placed in the negative pressure room.
- Identify those HDs in NIOSH Table 1 that do not require compounding prior to dispensing for patient use. NIOSH Table 1 HDs that only need to be counted or packaged may be included in the assessment of risk. For example, methotrexate tablets that are received and dispensed in unit-dose packaging, or a vial of methotrexate that will be dispensed intact to a patient for self-administration may be included in an organization’s assessment of risk, if alternative strategies are identified and implemented.
- Assess dosage forms of other HDs on the list to determine if they will be handled with all the precautions in USP <800> or if alternative strategies will be adequate to protect personnel. If an organization has the facilities to compound injectable cytotoxic agents, it is prudent to consider those containment strategies and work practices for non-Table 1 HDs and dosage forms. However, many HDs not found in Table 1 may have adequate hazard mitigation in place and can be considered for alternative strategies. Some organizations share their assessments of risk, but each organization needs to evaluate the specific practices at their own site when adding HDs and dosage forms to their assessment of risk.
- Review and document the assessment of risk at least annually. Note that new HDs and dosage forms must be included when they are first used, rather than waiting for the annual review.
Q: How can you ensure that safe handling practices are consistently and continuously followed?
A: In addition to assigning a Designated Person to oversee HD operations, training and requalification of personnel is one of the most important aspects of assuring safe practices for HD handling. Pharmacy staff must be adequately educated and must complete initial and requalification tests at the frequency defined in USP <800>, state regulations, accreditation organization standards, and health system policies. General training and requalification guidance is found in USP <795> for nonsterile compounding and USP <797> for sterile compounding. USP <800> requires inclusion of appropriate HD practices to be sure personnel are competent and proficient in the special needs of handling HDs.2
Q: What are the steps to developing policies and procedures for receiving, unpacking, and storing HDs?
A: Thorough policies and procedures (P&Ps) must begin from the point of delivery and then support the transfer of HDs to the ultimate storage/compounding area as quickly as possible. Receiving and unpacking are distinct issues. Receiving can occur in the regular receiving area, but the procedure for unpacking the HDs must include specific steps to limit the possibility of contamination. Include these key steps in your P&Ps:
Receiving
- Encourage suppliers to provide HDs in marked packages.
- Containers of Table 1 antineoplastics should arrive in separately labeled packages, identifiable as hazardous from the outside of the container, and the HDs should be enclosed in impervious plastic inside the tote or other packaging (see PHOTO).
- If you have a negative pressure area for receiving NIOSH Table 1 HDs, move the package to that location.

Unpacking
- Unpack the HDs that have been deemed entity-exempt in your assessment of risk and place them in their intended storage areas.
- Prepare the area for unpacking the NIOSH Table 1 antineoplastics and other HDs that you store with all the precautions listed in USP <800>.
- Check the integrity of the container. If it appears damaged, refer to your policy for handling damaged HD packages. USP <800> provides information concerning handling of damaged packages.
- Prepare the unpacking area, such as placement of a disposable chemo pad on the area where you will place the HD packages.
- Don one pair of gloves that meet standard ASTM D6978 (commonly called chemo gloves) and open the shipping container.
- Wipe the outside of the plastic bag containing the HDs with the method approved in your policy.
- Ideally, the HDs should remain in the plastic bag until the bag is opened in the negative pressure HD storage room. (This can be a separate room or can be inside the negative pressure buffer room or in the C-SCA.) If a pass-through chamber is part of your design, place the plastic bag containing the HDs in the pass-through chamber into the negative pressure room and open the bag in that room.
- If a pass-through is not included in your design, place the bag of HDs in an appropriate container (such as a dedicated plastic tote that can be decontaminated and cleaned) to transfer them into the negative pressure storage area.
Storage
- Place the HDs in their storage spot, preferably in plastic bins.
- Vials packaged in smooth coated cardboard may remain in their boxes, but corrugated cardboard or any other external shipping container should never be taken into an area used for sterile compounding.
Q: What are the advantages of HD wipe sampling?
A: Microbial environmental monitoring of PECs and SECs allow for the visualization of any contamination on growth media plates and must be completed for sterile compounding areas as described in USP <797>. Because HD contamination is not typically visible, wipe sampling serves to identify this risk. According to USP <800>, wipe sampling should be conducted in areas that are possibly contaminated with HD residue.2 The process allows pharmacy leadership to gain insight into staff exposure to HDs in places where these products are stored, compounded, and administered. Two types of systems are available for monitoring:
- Wipe sampling a defined area with a swab that is sent to a laboratory. The laboratory can detect a variety of antineoplastics or hormones. Results are reported back to the site within several days or weeks.3
- Wipe sampling a defined area with a swab that is placed in a solution and read via a reader specific to a single antineoplastic drug. Detection for three drugs (cyclophosphamide, doxorubicin, and methotrexate) is currently available. Results can be available within an hour.
There are merits to both types of systems. Reporting a panel of HDs is most useful for areas where multiple HDs are stored, compounded, or administered. Reporting a single agent is most useful for training, following spills, and when results are desired in the same time frame as potential contamination of the HD.4 Organizations handling HDs should recognize the risk of employee exposure to HDs and should incorporate adequate monitoring of areas where HD contamination is likely.5
 Patricia C. Kienle, RPh, MPA, BCSCP, FASHP, is director of accreditation and medication safety for Cardinal Health and is a member of the USP Compounding Expert Committee. Her comments herein are her own and not official information from USP.
Patricia C. Kienle, RPh, MPA, BCSCP, FASHP, is director of accreditation and medication safety for Cardinal Health and is a member of the USP Compounding Expert Committee. Her comments herein are her own and not official information from USP.
References
- US Centers for Disease Control and Prevention and National Institute for Occupational Safety and Health. NIOSH List of Antineoplastics and Other Hazardous Drugs in Healthcare Settings, 2016. www.cdc.gov/niosh/docs/2016-161/default.html. Accessed April 1, 2021.
- United States Pharmacopeial Convention. USP General Chapter <800> Hazardous Drugs–Handling in Healthcare Settings. www.usp.org/compounding/general-chapter-hazardous-drugshandling-healthcare. Accessed April 1, 2021.
- Villamaria RP. Buyer’s Guide: Wipe Analysis. Pharm Purch Prod. 2019;16(9),8.
- Polovich M, Ganio M, Kienle P. Best practices for monitoring surfaces for hazardous drug contamination: consensus conference recommendations and next steps. Presented at the Virtual 2020 ASHP Midyear Clinical Meeting & Exhibition. www.ashpadvantage.com/safesurfaces. Accessed May 25, 2021.
- Roussel C, Witt KL, Shaw PB, et al. Meta-analysis of chromosomal aberrations as a biomarker of exposure in healthcare workers occupationally exposed to antineoplastic drugs. Mutat Res. 2019;781:207-17.
Resource Box
Key References Detailing Safe Practices for HD Handling
- NIOSH Alert, 2004
National Institute of Occupational Safety and Health
- NIOSH List of Antineoplastic and Other Hazardous Drugs
National Institute of Occupational Safety and Health
- ONS Safe Handling of Hazardous Drugs, 3rd edition
Oncology Nursing Society
- Improving Safe Practice Handling for Hazardous Drugs
Joint Commission Resources, 2016
Like what you've read? Please log in or create a free account to enjoy more of what www.pppmag.com has to offer.








